Cancer is different for everyone. For the majority of patients, the diagnosis comes with far-reaching impacts on their lives - beyond the illness itself. However, suitable treatments can make a meaningful difference to patients’ lives - helping many live longer, manage symptoms more effectively, and maintain a better quality of life.
At Daiichi Sankyo Europe, we believe that true progress comes from listening to patients, understanding their perspectives, and ensuring that every advancement translates into real, significant benefits. Especially so in oncology, as a constantly evolving field that is driven by innovation and new scientific discoveries. We are deeply committed to advancing research and developing new treatment options to bring our therapies to any patient who can benefit.
Our Ambition: Improving Standards of Care in Oncology
With purpose, rigor and persistence, we aspire to create better tomorrows for people living with cancer and their loved ones. That is why we aim to maintain a high standard of innovation through continuous research and development of our technologies and our holistic approach to patient care.
Building on groundbreaking research from our labs in Japan, we are dedicated to advancing cancer treatment for solid tumours like breast, lung, and stomach cancer, as well as blood cancers. With 40 years of experience in oncology, we continuously aim to push the boundaries of science to develop new and innovative therapies. Since 2010, we have focused on developing ADCs.
We believe we are stronger together, uniting colleagues from diverse backgrounds and working as a trusted partner alongside healthcare providers, patient representatives, industry leaders, and more. Through openness and collaboration, we strive to bring new therapies to people living with cancer, because we know that only together, we can improve standards of care and create better futures through medicine.
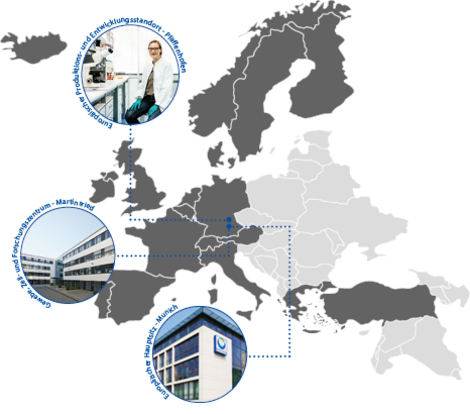
- Our European headquarter is located in Munich, Germany, with key operations across multiple sites. In Martinsried, our Translational Research Center Europe drives cutting-edge oncology research. Meanwhile, our state-of-the-art facility in Pfaffenhofen, opened in 2024, serves as a key manufacturing and distribution hub, supplying products to over 50 countries worldwide. Until 2030, we plan to invest around 1 billion euros into expanding Pfaffenhofen into a global innovation centre, reinforcing our commitment to advancing pharmaceutical development and manufacturing in Europe.
- Across Europe and Canada, we have 14 affiliates and aim to enlarge our footprint even further to maximise our impact for patients.

Compassion for Patients - Beyond the Medicines We Develop

We recognise that each patient is an expert in their personal disease and has a unique perspective about their care. This is why we actively listen to patients to understand their challenges and work to provide solutions that address their needs. We forge partnerships with different patient advocacy organisations and patient experts, to learn from real experiences and help ensure that patient needs are considered at the front and centre of our work.
Our Technology: Innovating for the Future of Cancer Therapies

Daiichi Sankyo has over 125 years of experience in researching and developing innovative medicines. This heritage inspires us to rise to new challenges. Since 2010, we have successfully focused on the development of antibody drug conjugates (ADCs) in oncology. ADCs are a highly targeted therapeutic approach, and we believe that our ADCs will significantly impact existing treatment options.
Whilst traditional therapies are effective in treating cancer cells, healthy cells may also be damaged due to their non-specific nature.1 This can lead to side effects which often impact patients’ quality of life.2
ADCs utilise a highly specific mechanism that helps efficiently target cancer cells with precision, reducing the risk of damage to healthy tissue.3 Daiichi Sankyo is continuously advancing this technology, striving to develop more targeted treatments for certain types of cancer. With around four million people diagnosed with cancer in Europe each year4, we are committed to contributing to pioneering treatment options where they are needed most.
Read more about our pioneering efforts in ADC technology.
Daiichi Sankyo Europe specialises across multiple types of cancer, committed to improve the standard of care in oncology and thus contributing to the enrichment of patients’ quality of life. Honing into the ADC technology, we aim to expand and develop our pipeline of ADC therapies - targeting more than 30 indications to reach nearly 400,000 patients worldwide.5
References
1. Tolossa Debela, D. et al. SAGE Open Med. 2021;12(9)
2. Yeo and Cannaday. Nursing: Research and Reviews. 2015;5(65)
3. Tsuchikama, K. et al. Nature Reviews Clinical Oncology. 2024;21(3)
4. European Cancer Statistics | Beat Cancer EU. [Accessed February 2025]
5. ENHERTU Business Briefing 2024 | Daiichi Sankyo. [Accessed February 2025]


